Food Safety
Four Reasons To Consider Rapid Microbial Screening of Beverage Products
When manufacturing your final product, the end goal is for your product to quickly pass quality control and get to market. However, traditional methods for microbial testing can take days or weeks for results and requires a number of manual processes, increasing the risk for technician error. In addition, many results are not quantitative and require visual inspection for interpretation. What if you could simplify this process with a rapid microbial screening method? How would getting rapid, quantitative results benefit your business and impact your bottom line? Keep reading to learn 4 reasons why you should switch to rapid microbial screening today.
1. One instrument for multiple beverage products
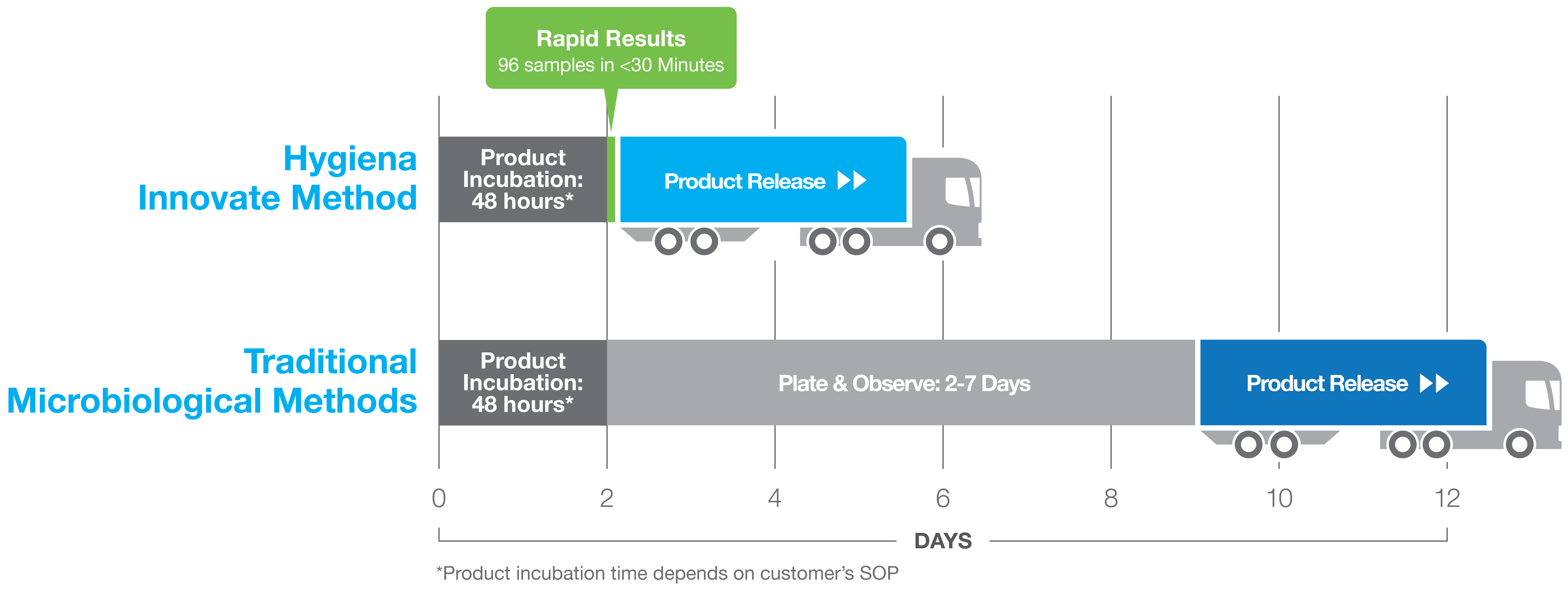
Do you have unique beverages that need testing? For laboratories supporting the beverage and dairy manufacturers, ATP testing is often done on the finished product to confirm the absence of microbial contamination. This requires sampling many individual product packages. Do you have a system that can handle all this? Rapid microbial screening is the solution. Rapid screening can provide reliable, reproducible results in typically 24-48 hours, resulting in significant savings. By testing and releasing the product in 2 days, you are saving hold times of two to 12 days, eliminating the need for large quarantine holding spaces. This not only shortens the hold cycle but also shortens the cash cycle so you can ship products sooner and get paid sooner. It also prevents hold-ups due to contamination that may occur in a batch, allowing you to address issues much more quickly.
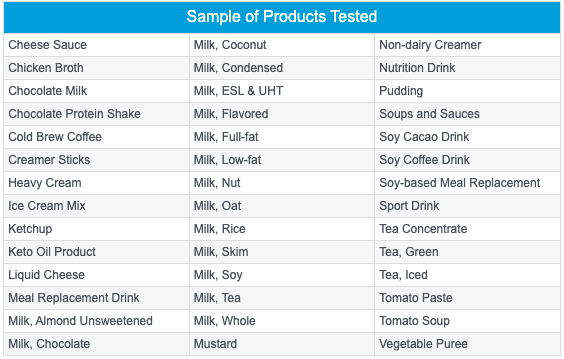
Of course, the rapid method must be consistent and reliable in order to ensure the quality and safety of your final product. This means testing products that are susceptible to microbial contamination by screening appropriate sample numbers before releasing them for distribution and sale to consumers. Fortunately, a well-validated adenosine triphosphate (ATP) rapid microbial screening method can do this. ATP is present in all living organisms, so can be used as an indicator of microorganism presence, even in highly processed dairy and beverage products. The screening is simple: product samples are treated with a proprietary reagent that reduces non-microbial sources of ‘background’ ATP. Then, a reaction is activated that causes a reaction with any existing microbial ATP, resulting in bioluminescence which can be read by a luminometer or light-recording instrument. When optimized, this allows for rapid screening of sample products quickly and accurately, detecting any potential contamination, allowing the vast majority of production to be released to market rapidly and efficiently. A great option for rapid microbial screening is The Innovate System which can be used to test a wide variety of product types - from cheese sauce to pulpy juices to thick puddings and even ketchup. The RapiScreen™ Beverage reagents designed for use with the Innovate System, have strong buffering capacities to neutralize high-acid content in juices and eliminate interference from complex ingredients in dairy formulations. No other rapid method covers such a wide range of products and applications. The Innovate System is flexible and easy to use, even with hard-to-test product types that are:
- Clear, opaque or highly pigmented
- Filterable or non-filterable
- Soluble or non-soluble
- High or low pH
- Preserved or non-preserved
- Aseptically packaged
Here are some of the matrices that have been tested: black tea, coffee concentrate, apple juice, orange juice, prune juice, oat-based juice, almond-based juice, tomato juice, and protein shakes. Below is the range of products and applications that Innovate System can test:
2. Results in as little as 24 hours
Why wait a week or more for results when you can have results in 24 hours?
We have worked with numerous customers to validate the detection of low levels of microbial contamination after 24 hours of incubation. Within 30 minutes, the RapiScreen™ kits can not only detect but also quantitate levels of microorganisms that are present - and 96 samples at a time. This shortens time to results and allows for quicker release of batches for consumer sale. This means you need less warehouse space, less holding space, and can easily batch testing for multiple sample types at once.
How can it detect contamination so quickly?
The Innovate platform features RapiScreen™, ATP bioluminescence technology, the industry standard for screening dairy products due to its speed and clear absence/ presence results. ATP testing eliminates the need for plate incubation, allowing product to be released days faster. The system can run 96 individual samples at once with no secondary incubation required. This is possible due to a unique technology that eliminates somatic cell (non-microbial) ATP and allows testing products with a wide range of pH values.
3. Over $500,000 in Cost Savings
You can reduce inventory and warehouse costs by releasing products days faster. With results in as little as 24 hours, the Innovate System can detect contamination in dairy, food, and beverage products where there is a low expectation of bio-burden such as in ultra-high temperature (UHT) or extended shelf-life (ESL) pasteurized products. After incubation, the Innovate System uses a simple three-step process for testing samples which makes test set up easy and reduces technician time.
Following product analysis (less than 30 minutes), results are automatically exported to the onboard database and a product report is generated for all samples tested, allowing rapid release of products testing negative. This allows for quickly reducing inventory as products can be shipped to market. Overall, it also decreases the manufacturing costs - as releasing product sooner, means more revenue sooner, and faster movement of more product through manufacturing - more volume released at the same or less cost. By shortening time to results, facilities can reduce inventory requirements, warehouse space and safety stock, resulting in significant savings, which impacts the bottom line positively. In fact, our studies have shown that for over 200 installations in over 65 countries, average dairy industry customers have saved over $500,000 in the first five years of implementation. The same should be true for beverage customers who use the Innovate System.
4. High Volume can be automated
While the Innovate System can help shorten time to results, improve inventory movement and save technician time, large sample volumes can still take time to analyze. Therefore, for large sample volumes, the testing process can be further streamlined with the use of the Innovate Autosampler III. It streamlines product sampling from any style carton to the testing plate, increasing efficiency and consistency. It also has the added flexibility of selecting the number of channels for sampling the finished product (from 4 - 8) to ensure maximum sampling efficiency.
The Innovate Autosampler III offers a continuous workflow to eliminate any sampling interruptions and can prepare 2,000 samples per hour with the ability to hold 96-well plates in a queue for subsequent testing on the Innovate System. Maintenance and downtime are also minimized with on-board controls and the integrated software allows communication between the production line and the Innovate System using existing LIMS functionality.
The Bottom Line
Implementing a rapid, easy-to-use solution for initial microbiological testing aids in the analysis of the final beverage or milk product for contamination. The Innovate System can deliver quality control results in less than 30 minutes, allowing beverage and dairy manufacturers to rapidly confirm the quality of their final products and release them quickly to market. Contact us to talk about the Innovate Beverage Kit capabilities and how they can be implemented at your facility.