Food Safety
IFTPS 2026: The future of aseptic food & beverage production is already taking shape
The aseptic food and beverage industry is entering its next phase of evolution.
The aseptic food and beverage industry is evolving rapidly, with automation, connected data systems, and faster microbial testing reshaping how producers manage risk and release products. Insights from the Institute for Thermal Processing Specialists Annual Meeting 2026 in Atlanta highlight a clear shift toward scalable, validated solutions that enable earlier contamination detection, reduce product hold times, and support more confident commercial sterility decisions.
The aseptic food and beverage industry is entering its next phase of evolution.
A few themes stood out across discussions with producers, scientists, and processing authorities:
-
Automation is no longer optional
From sampling to decision-making, the direction is clear: systems will continue to move toward full automation to improve consistency, reduce labor dependency, and accelerate release timelines.
-
Industry 5.0 is becoming a reality
The combination of human expertise and AI-driven insights is how food safety decisions are made. Faster data interpretation and more confident decisions are now a competitive advantage.
-
Rapid methods are gaining ground, but adoption still requires trust
While there is strong interest in technologies such as ATP-based rapid microbial detection, validation, and alignment with processing authorities remains critical to wider adoption.
-
Collaboration is key to progress
From manufacturers to technology providers and regulatory bodies, advancing aseptic production requires a shared approach to validation, standardization, and education.
At Hygiena®, these conversations reinforce the direction we are already focused on:
supporting producers with faster, validated, and scalable approaches to commercial sterility and environmental monitoring.
What this means in practice for aseptic producers
The themes emerging from the Institute for Thermal Processing Specialists are not theoretical. They reflect real operational pressures already being felt across aseptic dairy and beverage production.
Automation is solving more than labor challenges
Automation is often framed as a response to labor shortages, but its impact extends beyond that.
In aseptic environments, automation is increasingly being used to:
-
Standardize sampling across incubation windows
-
Reduce variability introduced by manual handling
-
Increase throughput without increasing headcount
-
Enable earlier, more consistent decision points
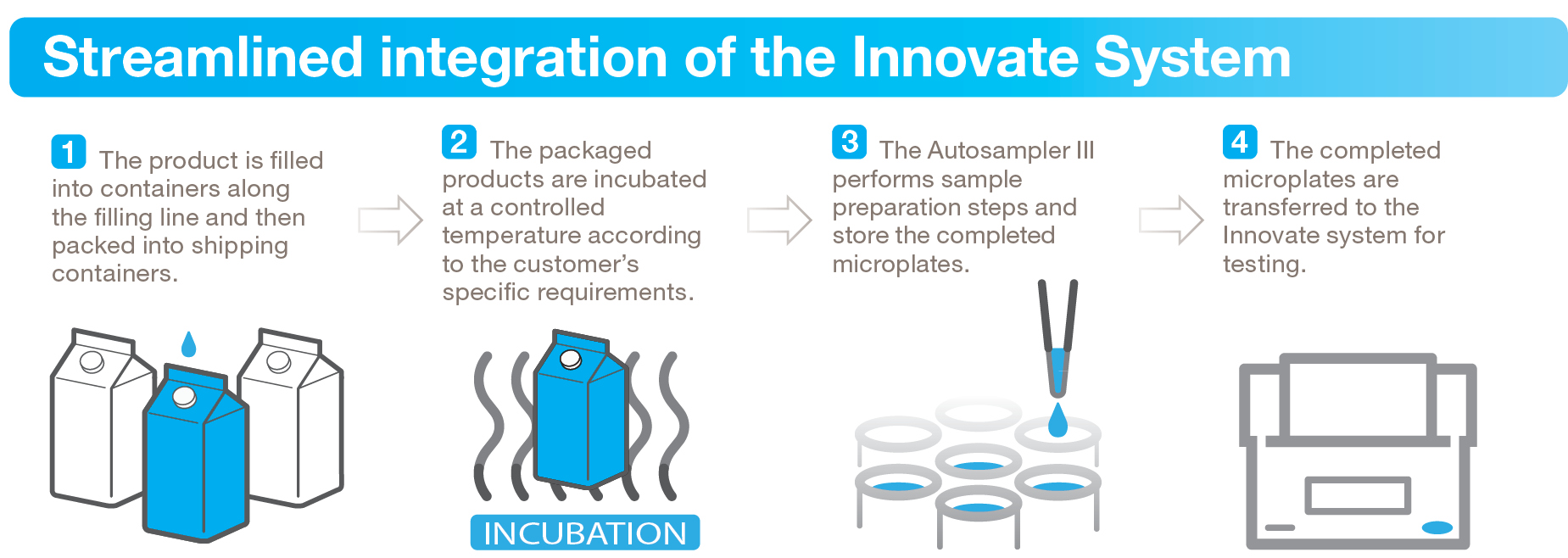
For producers managing high sample volumes, this shift is enabling a move away from batch-based constraints toward continuous, scalable testing workflows.
Solutions that combine automated sampling with rapid detection methods are becoming central to this transition.
Explore how automation supports microbial testing workflows:
https://www.hygiena.com/instruments-and-automation/monitoring-systems/innovate
Faster data is reshaping release decisions
One of the clearest shifts discussed at IFTPS is the expectation of earlier insight into microbial status.
Traditional methods introduce delays that can:
-
Extend product hold times
-
Increase storage and inventory costs
-
Limit responsiveness to contamination events
Rapid microbial screening technologies, particularly those based on ATP bioluminescence, are enabling:
-
Earlier indication of microbial presence
-
Shortened time to decision for commercial sterility
-
Reduced reliance on retrospective testing
For UHT, ESL, and aseptic beverage producers, this is not just a quality improvement; it is a commercial advantage.
Learn how rapid microbial detection supports faster release decisions:
https://www.hygiena.com/invest-in-innovate
Understand the role of testing in commercial sterility:
https://www.hygiena.com/food-safety/microbial-contamination/commercial-sterility-testing
Validation remains the critical bridge to adoption
Despite strong interest in rapid methods, a consistent message at IFTPS was that adoption depends on confidence.
This confidence is built through:
-
Product-specific validation on real matrices
-
Alignment with internal quality teams and process authorities
-
Clear demonstration of equivalence or advantage vs traditional methods
Producers are increasingly seeking partners who can support this process, not just provide technology.
See how Hygiena supports method validation and real-world application:
https://www.hygiena.com
Connected systems are becoming the standard
As Industry 5.0 concepts take hold, testing is no longer viewed in isolation.
There is a growing expectation that systems should:
-
Integrate data across hygiene monitoring, environmental testing, and finished product quality
-
Provide centralized, audit-ready reporting
-
Enable trend analysis and proactive risk management
Explore connected data and analytics for food safety:
https://www.hygiena.com/suretrend-data-analytics
This shift toward connected data ecosystems is enabling producers to move from reactive testing to predictive quality management.
How Hygiena is supporting this shift
The direction discussed at the Institute for Thermal Processing Specialists aligns closely with Hygiena’s approach to aseptic production environments.
Across the production lifecycle, solutions are designed to support:
-
Automated, high-throughput microbial screening for finished product quality
-
Rapid hygiene and environmental monitoring to verify cleaning effectiveness
-
Validated pathogen detection workflows for risk mitigation
-
Integrated data management platforms for visibility and compliance
Explore Hygiena’s full dairy and aseptic testing portfolio:
https://www.hygiena.com/dairy-safety-testing
Looking ahead
The aseptic food and beverage industry is not waiting for the future, it is actively building it.
The conversations at Institute for Thermal Processing Specialists make it clear that success will depend on:
-
Speed of insight
-
Confidence in results
-
Ability to scale without compromise
For producers, the question is no longer if these changes will happen, but how quickly they can be implemented to remain competitive.